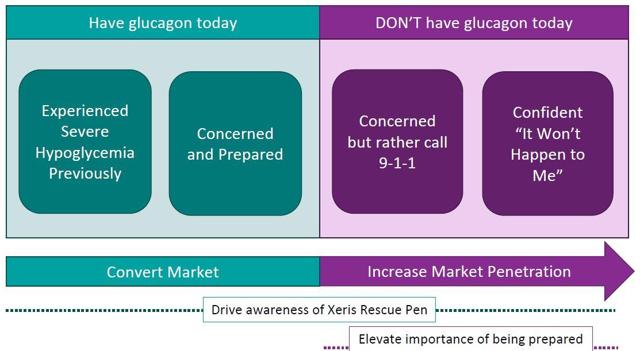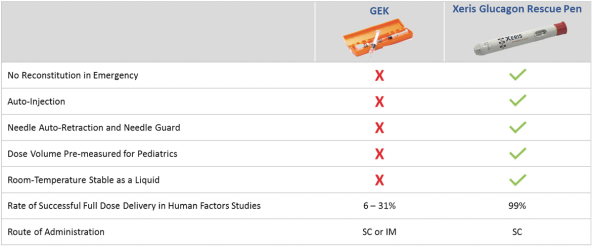
Xeris Pharmaceuticals Receives European Commission Approval of Ogluo™ ( glucagon) Injection for the Treatment of Severe Hypoglycaemia in Adults, Adolescents, and Children Aged 2 Years and Over With Diabetes Mellitus | Business Wire

Next-Gen Emergency Glucagon: Xeris to the Rescue? | Diabetes technology, Diabetes diet plan, Diabetes management